
ResearchResearch
1.A.5 (b) AppA
AppA is a light and redox-responding regulator of photosynthesis gene
transcription in Rb. sphaeroides, where it can be found in two different
functional forms. Under anaerobic low-light growth conditions, AppA is
in a “dark adapted” form, which is able to bind and inactivate the repressor
PpsR, thus allowing the RNA polymerase to maximally transcribe photosynthesis
genes. Under aerobic highlight conditions or under strong blue light illumination,
FAD in AppA is photoexcited and AppA is transformed into a signaling state
(“light-adapted” form) that is incapable of interacting with the photosynthesis
repressor PpsR. Under these conditions, there is a maximal repression of
the photosynthesis gene expression.
The photoreaction kinetics of the BLUF domain of AppA5-125 was studied
by monitoring time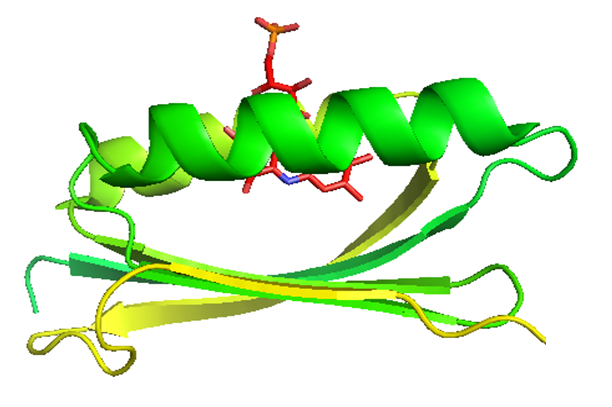 dependence of an apparent diffusion coefficient (D) using the pulsed laser
induced transient grating technique. It was found that D of the photo-product
is time dependent. From the concentration dependence of the reaction rate,
it was concluded that the BLUF domain of AppA forms a dimer upon the photoexcitation.
Since AppA exists as a dimeric form in the ground state, this dimerization
reaction indicates the tetramer formation in the signaling state. From
the slope of the plot of observed rate constants (kobs) against the AppA
concentration, the second order rate constant is determined to be ~2.5×105
M-1s-1, which is about 4 orders in magnitude lower than the diffusion controlled
reaction. It indicates that a relative orientation of the protein molecules
during the dimerization process causes additional constraints, which slow
down the reaction rate.
dependence of an apparent diffusion coefficient (D) using the pulsed laser
induced transient grating technique. It was found that D of the photo-product
is time dependent. From the concentration dependence of the reaction rate,
it was concluded that the BLUF domain of AppA forms a dimer upon the photoexcitation.
Since AppA exists as a dimeric form in the ground state, this dimerization
reaction indicates the tetramer formation in the signaling state. From
the slope of the plot of observed rate constants (kobs) against the AppA
concentration, the second order rate constant is determined to be ~2.5×105
M-1s-1, which is about 4 orders in magnitude lower than the diffusion controlled
reaction. It indicates that a relative orientation of the protein molecules
during the dimerization process causes additional constraints, which slow
down the reaction rate.
(Back)


photo-physical-chemistry lab,京都大学大学院理学研究科 化学専攻 光物理化学研究室
〒606-8502
Kitashirakawaoiwakecho
Sakyoku, Kyoto, Japan
TEL +81-75-753-4026
FAX +81-75-753-4000
<Links for members>
Bake Web mail (Set up)
Manuals