Research Outline
Creation of novel inorganic-organic hybrid materials using atom, electron, ion, and spin degrees of freedom
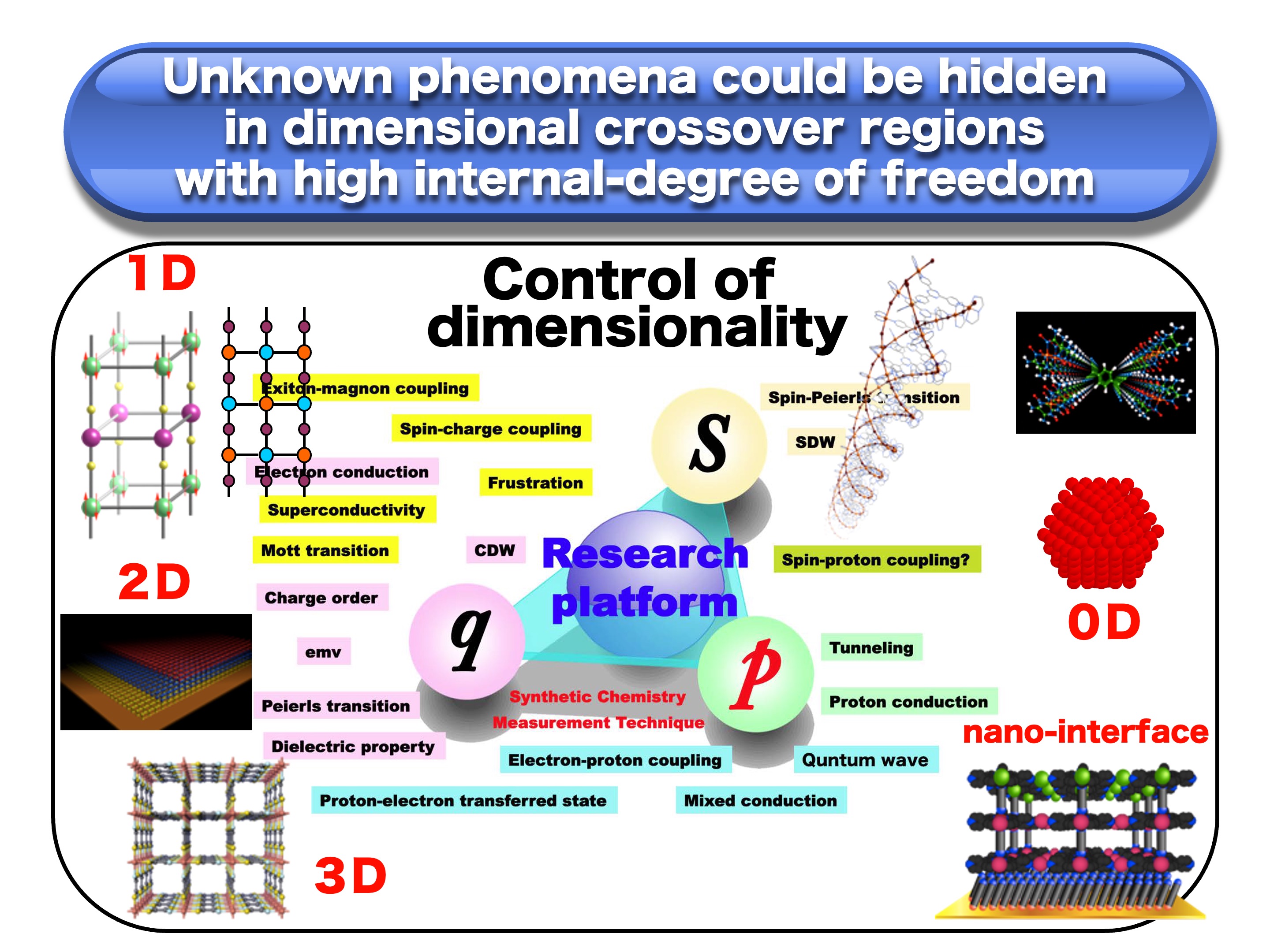
1) Creation of novel quantum phases of electrons, ions, and spins in dimensional crossover regions
In the 21st century, the emergence of new molecular devices with flexibility and diversity, such as those found in biological functions, is increasingly expected. Although the basic concept for the realization of these new molecular devices is still being explored, we are aiming at the flexible construction and control of quantum-mechanical electronic (superconducting, ferromagnetic, ferroelectric, metallic, insulating, etc.) and ionic phases (especially super-protonic conduction, proton tunneling, and protonic quantum-paraelectric phases). By combining the diversity of electronic states of metal ions and the diversity of designability of organic ligands, we aim to create new materials with unique crystal and electronic structures, and to develop new functionalities and properties based on the quantum size effect, nonlinear electrical conduction, nonlinear optical effect, dielectric response, and various fluctuation effects.
The goal is to establish the basis for the realization of "molecular devices" that are similar to biological functions. Our research targets are not limited to inorganic and organic materials, but include low-dimensional strongly correlated electrons systems, mixed-valence materials with strong negative U-interactions, hydrogen-bonded charge-transfer complexes, electrically conductive coordination polymers, organic superconductors, organic spin liquids, etc., which are located in dimensional crossover regions (Figure 1).
2) Establishment of protoelectronics
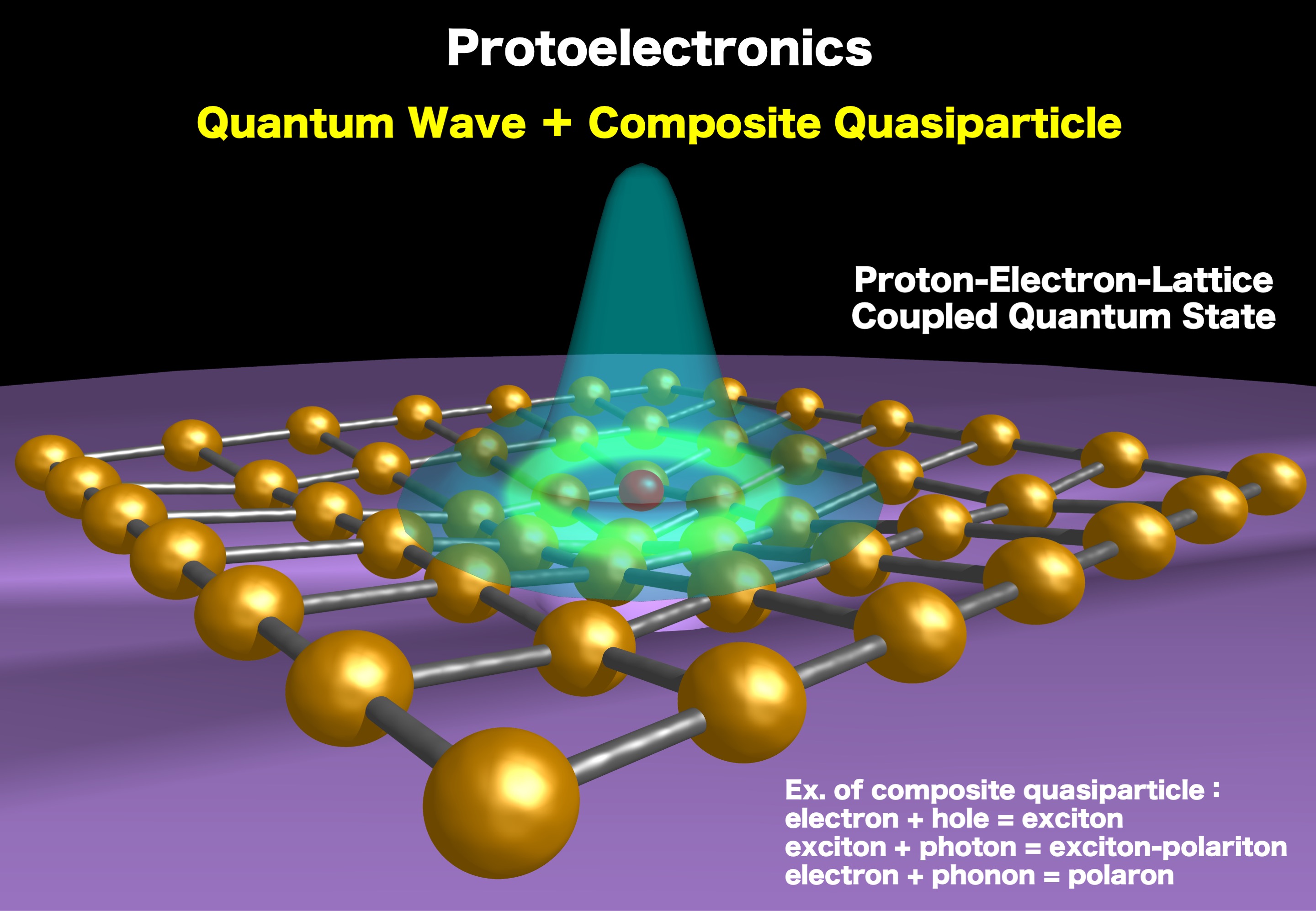
Solid-state protonics is defined as the comprehensive science and technology of freely manipulating hydrogen in solids. Electronics, which is the science and technology of manipulating electrons in solids, has almost been established, as seen in semiconductor technology, and it is possible to control the holding and moving of electrons almost at will, from insulators to semiconductors, metals, and superconductors. On the other hand, the freely manipulating of hydrogen in solids has not yet been achieved, and the establishment of solid-state protonics as an academic field is a challenge for the 21st century.
Since hydrogen is the lightest element and has quantum wave properties, if the degrees of freedom of hydrogen (proton, protium, hydride, nuclear spin, etc.) can be used in addition to the degrees of freedom of electrons (electron, hole, polaron, bipolaron, electron spin, etc.), a new fusion technology with more freedom and flexibility, "Protoelectronics" will be born. Hydrogen is composed of one proton and one electron, making it the simplest and lightest of all the elements. Hydrogen has only one electron, so electron correlation and many-body problems, such as electron repulsion effects, can be ignored. However, it is difficult to manipulate hydrogen freely in the solid state because of its relatively large electronegativity and the relatively strong binding of the 1s electron to the nucleus, in addition to its less itinerant nature compared to electrons. However, if we focus on the fact that hydrogen can take a continuous charge from -1 to +1 due to its moderate electronegativity, we can control hydrogen by coupling it with the degrees of freedom of the electron and lattice systems (Figure 2). In other words, depending on the strength of the covalent or ionic bond, hydrogen can be freely changed into hydride ion (H-), protium (H-), proton (H+), and so on. The charge state of hydrogen can be freely verified depending on the electron-donating or electron-accepting properties of the counter partner. “The softer the electronic system, the easier it is to handle hydrogen.”
In our laboratory, we have been studying various electronic phase transitions based on spin-charge-lattice strong coupling, quantum electron-proton coupling, super-proton conductivity in coordination polymers, mixed conductivity of electron and proton), and quantum properties induced by proton beam irradiation.
3) Elements strategy with multi-element nano-alloys and exploration of unknown materials by materials-creation informatics with HTS

In the world, there are many combinations that do not mix no matter how hard we try, like the relationship between water and oil. For example, iron and copper are two elements that do not mix at the atomic level. In fact, most of the alloy catalysts used in society are composed of elements that do not mix at the atomic level, and do not maximize the synergistic effect between different metallic elements. Since there are about 50 stably available metallic elements, there are about 1,200 combinations of them in binary alloys. Of these, less than 30% can be mixed at the atomic level in any proportion (all proportional solid-solution alloys), which means that human beings have not yet been able to utilize more than 70% of the alloy combinations.
In our laboratory, we are promoting research on the creation of new materials by mixing immiscible metallic elements, which have a relationship with water and oil, at the atomic level. One of our research strategies is that in solid-solution alloys, in which the constituent elements are randomly and uniformly mixed at the atomic level, it is possible to continuously control the electronic state, i.e., the function and physical properties, depending on the composition ratio. Therefore, if we can build a technology to freely mix and manipulate all kinds of elements, it will be possible to create new elements that surpass natural elements by combining the target element with other elements. Recently, the development of multi-element nanoalloys (Figure 3), in which arbitrary elements are mixed in arbitrary proportions, is about to be realized using a continuous-flow solvothermal non-equilibrium nanoalloying process that we have specially developed. Based on this science and technology, high-performance hydrogen storage materials, solid catalysts, electronic materials, optical materials, and magnetic materials are being developed through materials creation informatics with high-throughput screening (HTS).
